Standard Trinity
The “Standard Trinity”: A Unified Foundation
Section titled “The “Standard Trinity”: A Unified Foundation”The true power of digital health is realized when DICOM, FHIR, and SNOMED CT are integrated:
- Imaging (DICOM) provides the visual evidence.
- Exchange (FHIR) provides the structural pipe.
- Vocabulary (SNOMED CT) provides the semantic clarity. Together, they ensure that a patient’s health record is not just digital, but semantically rich, longitudinal, and universally understandable.
The Data Transformation Journey
Section titled “The Data Transformation Journey”As illustrated in Figure 1, the journey from raw clinical text to a secured, interoperable record requires a rigorous transformation that maps human-readable notes to global standards.
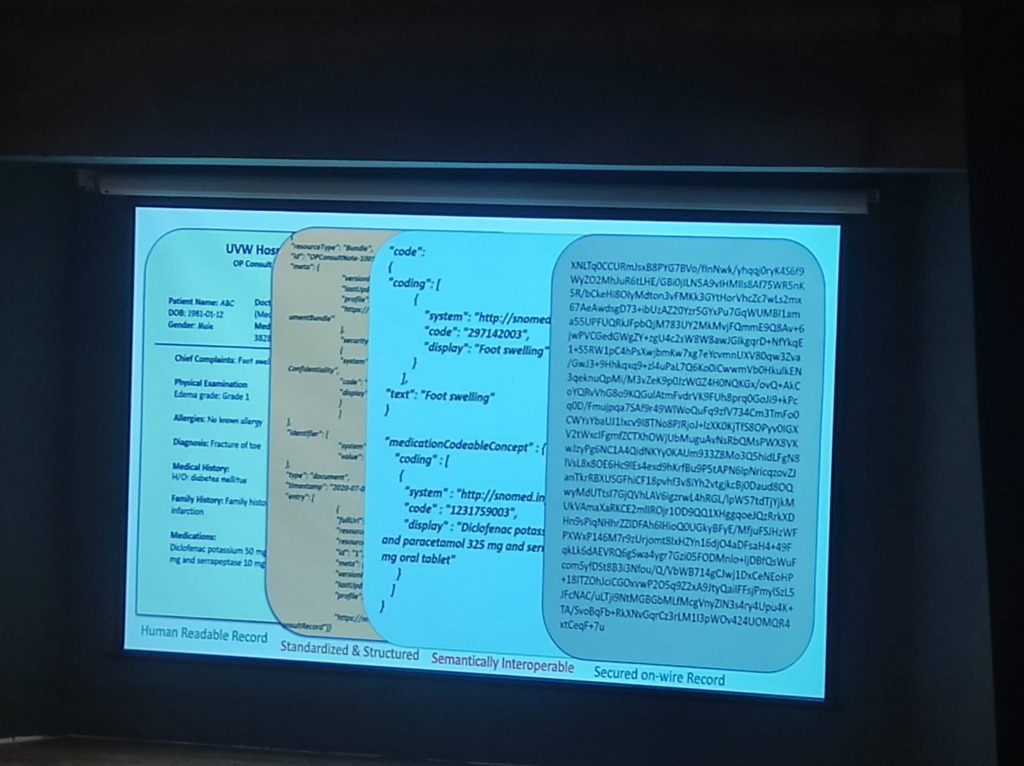 Figure 1: The Data Transformation Journey from human-readable records to semantically rich, secured data.
Figure 1: The Data Transformation Journey from human-readable records to semantically rich, secured data.
- Human Readable Record: The raw clinical input (e.g., “EDEMA grade 1”, “Diclofenac Potassium 50mg”).
- Standardized & Structured: Formatting the data into machine-level bundles (JSON/XML).
- Semantically Interoperable: Mapping terms to global standards like SNOMED CT (e.g., Code “297142003” for Foot swelling).
- Secured on-wire Record: Applying encryption to ensure data is protected as it moves across the network.
FHIR: The Blueprint for Exchange
Section titled “FHIR: The Blueprint for Exchange”FHIR (Fast Healthcare Interoperability Resources) is the global standard developed by HL7 International that enables this exchange. Crucially, the Ayushman Bharat Digital Mission (ABDM) uses FHIR as its primary data structure for creating India’s unified health infrastructure.
 Figure: NRCeS maintains the official FHIR Profiles and Implementation Guides tailored for the ABDM ecosystem.
Figure: NRCeS maintains the official FHIR Profiles and Implementation Guides tailored for the ABDM ecosystem.
-
NRCeS Stewardship: As detailed by Ms. Manisha Mantri (NRCeS), the National Resource Centre for EHR Standards (NRCeS) maintains the FHIR Profiles and Implementation Guides tailored for ABDM.
-
Comprehensive Specs: Combining a data dictionary, discrete objects/structures, and value tables for terminology.
-
Modular Resources: Providing over 145 standard structures covering Healthcare Entities (Patient, Practitioner), Clinical Information (Condition, Procedure), and Financial Information (Claims, Invoice).
-
Reusable & Extensible: FHIR is designed to be customized for specific use cases through extensible Profiles.
-
ABDM FHIR R4 Usage Samples: To accelerate integration with the Ayushman Bharat Digital Mission, official usage samples are provided for both Java and .Net environments.
-
Mandatory FHIR R4: The Integration Baseline for the ABDM ecosystem.
The Big Tech Paradox: The Apple Case
Section titled “The Big Tech Paradox: The Apple Case”- The Compliance Question: A common question in digital health circles is why global giants like Apple are not ABDM compliant.
- The Standards Gap: While Apple Health provides a sophisticated personal health record platform, it does not currently adhere to the mandatory HL7 FHIR R4 profiles finalized by the NRC for India.
- Proprietary vs. Open: Big Tech’s reliance on proprietary data models creates a friction point with national missions that mandate open, interoperable standards for public-private data exchange.
DICOM: Decoding Medical Imaging
Section titled “DICOM: Decoding Medical Imaging”While FHIR handles the clinical and administrative data, DICOM (Digital Imaging and Communications in Medicine) is the global specification for exchanging medical images.
 Figure: DICOM standardizes the output of medical imaging hardware preventing vendor lock-in.
Figure: DICOM standardizes the output of medical imaging hardware preventing vendor lock-in.
- Unified Imaging: Standardizing output from hardware like X-Ray, CT, and MRI machines to ensure seamless communication with PACS (Picture Archiving and Communication Systems).
- Clinical Inference through Metadata: Capturing critical attributes like laterality (left vs. right) and modality (CT vs. MRI), which are foundational for building advanced clinical inference models.
- DICOM & FHIR Synergy: The two standards work in tandem; for example, a FHIR DiagnosticReport resource can reference a DICOM ImagingStudy while exchanging information.
SNOMED CT: The Clinical Vocabulary
Section titled “SNOMED CT: The Clinical Vocabulary”While DICOM captures the pixels and FHIR captures the structure, SNOMED CT provides the meaning.
Facing the “Semantic Chaos”
Section titled “Facing the “Semantic Chaos””To illustrate the challenge of synonymy in clinical documentation, participants contributed the various ways they’ve seen common tests recorded in different hospital systems.
 Figure: Participant-Generated Word Cloud: The dozens of ways “Blood Sugar” is recorded—illustrating why a unified vocabulary like SNOMED CT is the only path to a longitudinal record.
Figure: Participant-Generated Word Cloud: The dozens of ways “Blood Sugar” is recorded—illustrating why a unified vocabulary like SNOMED CT is the only path to a longitudinal record.
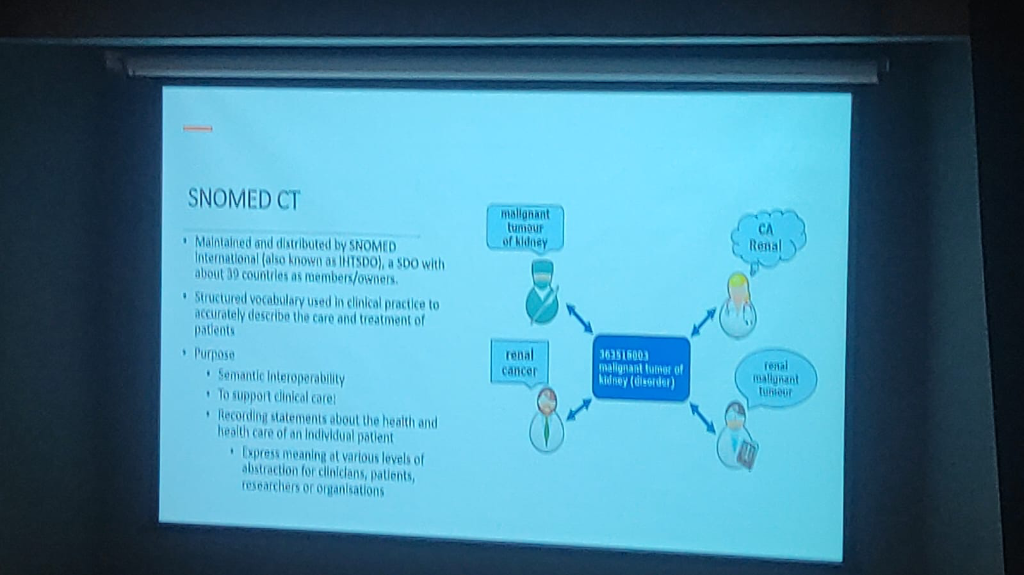 Figure: SNOMED CT maps multiple clinical terms to a single concept identifier, ensuring semantic interoperability.
Figure: SNOMED CT maps multiple clinical terms to a single concept identifier, ensuring semantic interoperability.
- Unified Meaning: Providing a structured vocabulary that accurately describes clinical care—for example, resolving terms like “Renal Cancer” and “Malignant tumor of kidney” to a single code (363516003).
- Automated Semantic Mapping: Enabling SNOMED CT and ICD to be mapped automatically, allowing clinicians to document with clinical precision while administrative requirements are handled in the background.
- Semantic Interoperability: Ensuring that data remains consistent and understandable across different systems, clinicians, and researchers.
- Multidimensional Abstraction: Expressing clinical meaning at various levels to support both precise detail and high-level population health analysis.
Advanced Analytics through the SNOMED CT Data Model
Section titled “Advanced Analytics through the SNOMED CT Data Model”Beyond simple terminology, SNOMED CT provides a robust Data Model that simplifies complex clinical analytics.
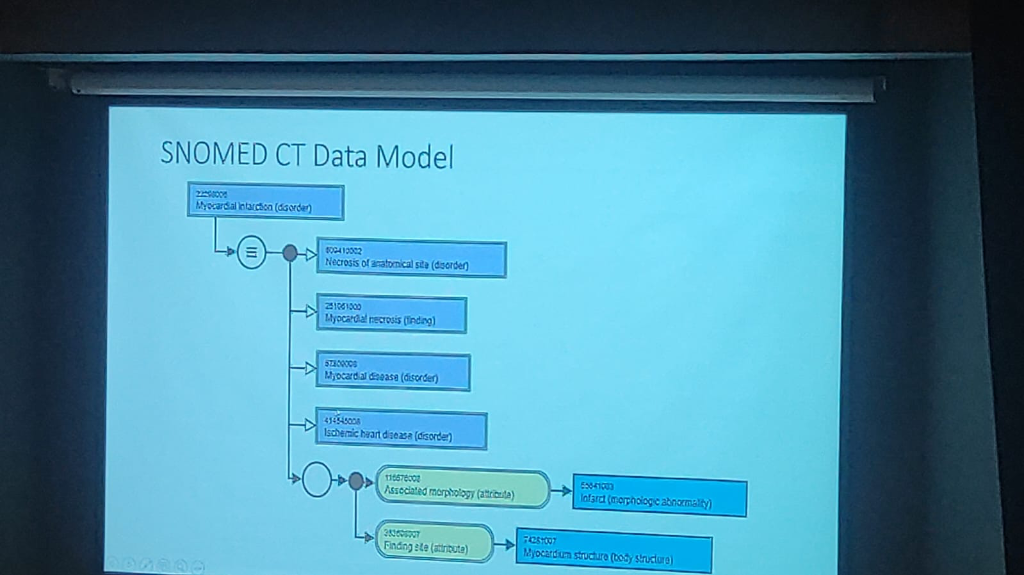 Figure: The SNOMED CT Data Model facilitates automated inference by linking conditions to their morphological and anatomical attributes.
Figure: The SNOMED CT Data Model facilitates automated inference by linking conditions to their morphological and anatomical attributes.
- Relationship Mapping: The model enables associated disease analysis by linking concepts through specific attributes like Associated Morphology (e.g., Infarct) and Finding Site (e.g., Myocardium structure).
- Automated Inference: By structuring clinical data into logical hierarchies (e.g., Myocardial infarction → Necrosis of anatomical site → Myocardial necrosis), it allows for automated inference and more accurate epidemiological research.
- Data Consistency: This machine-level data model ensures that data points from diverse sources can be aggregated and analyzed without manual normalization, making large-scale clinical trials and public health surveillance far more efficient.
LOINC: Standardizing Lab Observations
Section titled “LOINC: Standardizing Lab Observations”While SNOMED CT handles clinical findings, LOINC (Logical Observation Identifiers Names and Codes) is the universal standard for identifying medical and laboratory observations.
 Figure: LOINC provides distinct codes for identifying specific lab tests, ensuring comparability across different laboratories.
Figure: LOINC provides distinct codes for identifying specific lab tests, ensuring comparability across different laboratories.
- Laboratory Precision: It details the exact method, components, and systems used for a particular test (e.g., Code 9156-0 for Glucose in Urine by automated test strip).
- Universal Coding: LOINC ensures that a “Glucose” test from one lab is understood identically by any other system, regardless of the local equipment used.
ICD: Disease Classification & Reporting
Section titled “ICD: Disease Classification & Reporting”Maintained by the World Health Organization (WHO), ICD (International Classification of Diseases) is the global standard for health data, clinical documentation, and aggregation.
 Figure: ICD allows for the tracking of disease incidence and supports evidence-based health policy.
Figure: ICD allows for the tracking of disease incidence and supports evidence-based health policy.
-
Public Health Mandate: ICD codes are mandated for monitoring the incidence and prevalence of communicable diseases and for accurate mortality reporting.
-
Epidemiological Insights: It allows government bodies to conduct epidemiological research on health trends and compare data across different periods and locations.
-
National Identifiers (ABHA): The common thread bridging fragmented medical records into a longitudinal national history.
-
The SOP Gap: Digitization must focus on SOPs (Standard Operating Procedures), which are currently lacking across the board.
-
The Build vs. Buy Dilemma: A core strategic question for any institution is whether to build from scratch (custom design and development), buy COTS (Commercial Off-The-Shelf) applications, or adopt a hybrid approach of buying and customizing.
-
Phased Implementation: To minimize “teething problems,” adoption must be phased and well-planned rather than a “big bang” rollout.
C-DAC National Toolkit Suite (Speaker: Ms. Manisha Mantri)
Section titled “C-DAC National Toolkit Suite (Speaker: Ms. Manisha Mantri)”The Centre for Development of Advanced Computing (C-DAC) provides a comprehensive Medical Informatics SDK Suite to support national standardization:
- CSNOtk: C-DAC’s Toolkit for SNOMED CT, enabling standardized clinical terminology across all electronic records.
- CLNtk: C-DAC’s Toolkit for LOINC, standardizing laboratory and clinical observations.
